A Nine-Year-Old Boy with Lumps at the Base of the Fingers
August 17, 2016
A nine-year-old boy reported lumps at the base of the middle finger on each hand. He had been diagnosed with osteochondromatosis, and lesions involving the right great toe and the left tibia had been excised at one and two years of age, respectively. Osseous protrusions had later been observed on the right tibia and the left third toe at five and six years of age, respectively. Radiographs revealed periarticular calcification at the base of the proximal phalanx of both middle fingers and osteochondromas on the middle phalanx of the left middle finger and the metacarpus of the left small finger (Fig. 1). These were left untreated as they did not disturb activities of daily living. One year after his previous visit, at the age of nine years, radiographs showed that the osteochondromas on both middle fingers had increased in size and pointed toward the adjacent joints.
The lesions on the middle and proximal phalanges of the left middle finger were subsequently excised at the age of ten years and seven months because they caused pain and tenseness in activities of daily living (Fig. 2). However, the osteochondroma on the right middle finger was left untreated (Fig. 3-A). Radiographs made at eleven years and six months of age showed that the lesion on the right middle finger, which had not been treated surgically, had begun to decrease in size (Fig. 3-B), and it was mostly resolved at the age of twelve years and nine months (Fig. 3-C). In contrast, the base of the proximal phalanx of the middle finger was still bulky. The osteochondroma on the metacarpus of the left small finger also spontaneously resolved. Meanwhile, a new osteochondroma arose on the ulnar side of the bottom of the proximal phalanx, while the osteochondroma on the opposite side resolved spontaneously (Fig. 3-C). At the age of seventeen years and one month, radiographs showed that the lesion on the right middle finger was completely resolved and the base of the proximal phalanx was restored in terms of shape (Fig. 3-D). The new osteochondroma on the ulnar side of the bottom of the proximal phalanx also resolved.
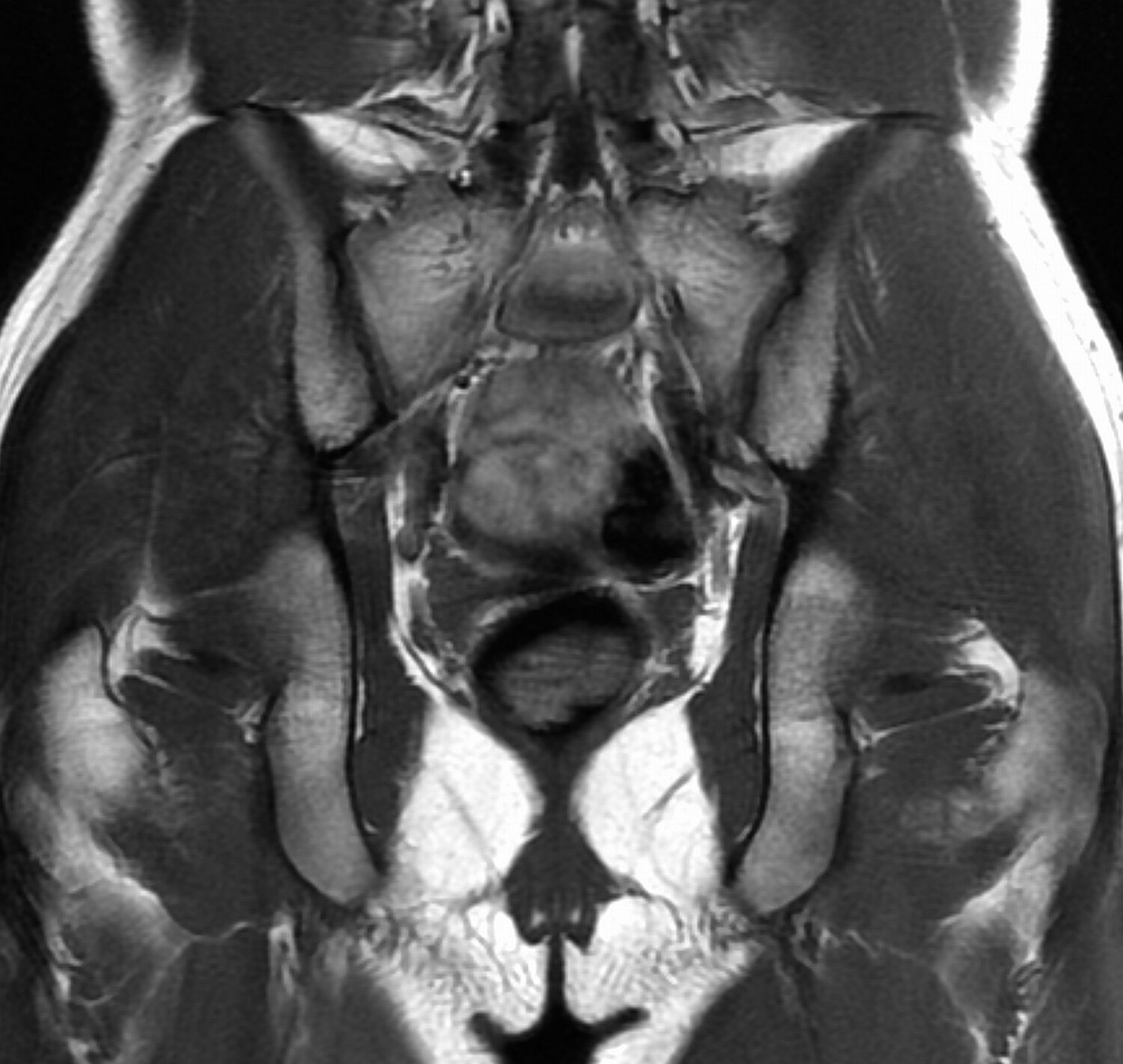
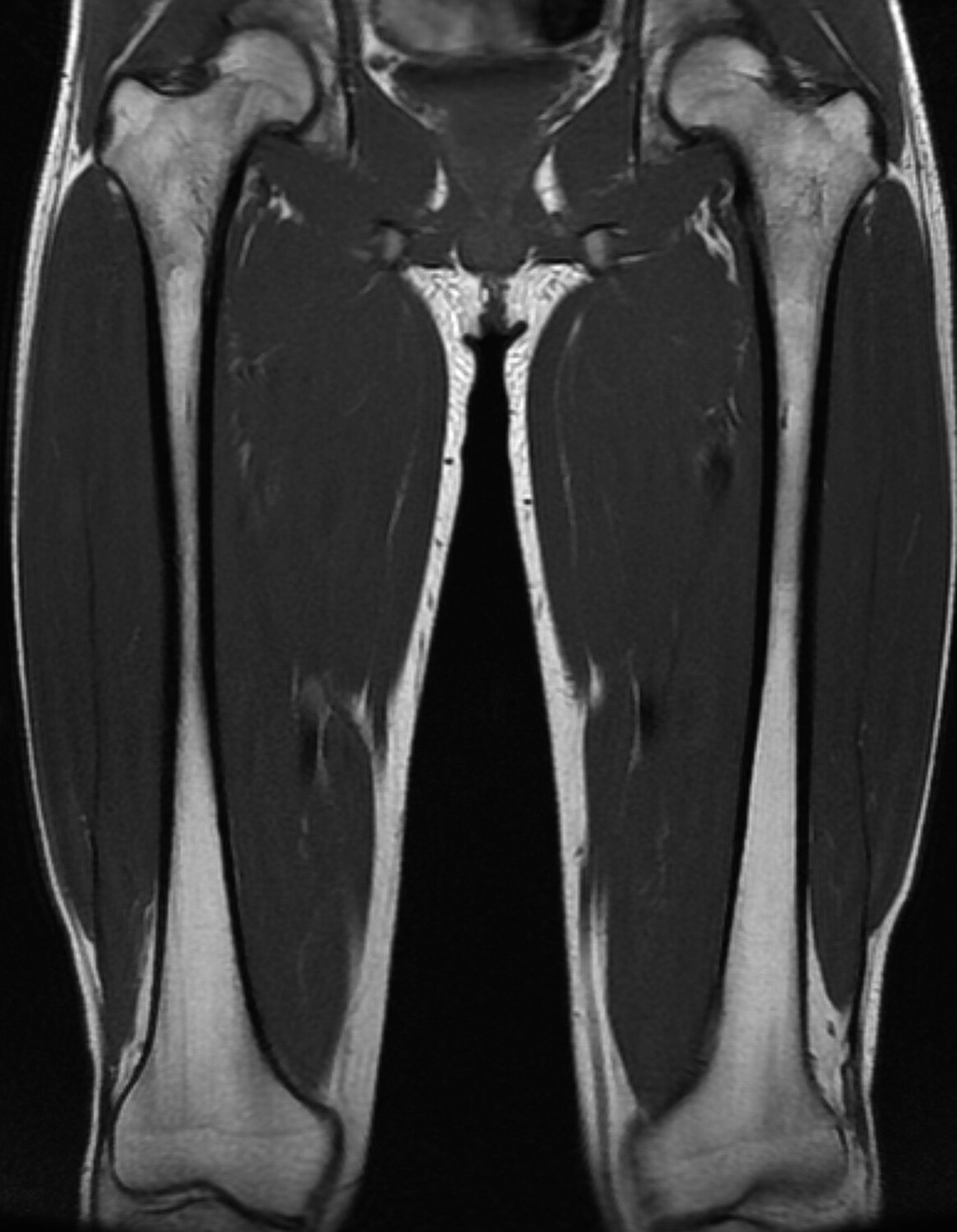
A radiographic survey of the skeleton failed to identify any enchondromatous lesions (Fig. 4). MRI (magnetic resonance imaging) also failed to reveal the presence of any enchondromatous pelvic or proximal femoral lesions (Fig. 5 and Fig. 6). The family history revealed that the patient’s mother had also been affected on both hands in her youth, although her tumors resolved spontaneously.
RT-PCR (reverse transcription-polymerase chain reaction) analysis was performed at the Department of Medical Genetics, Tohoku University School of Medicine, to detect genetic mutations. A heterozygous mutation in PTPN11 (PTPN11c.1093-1G>A), a genetic cause of metachondromatosis, was identified in peripheral blood.
Proceed to Discussion >>Reference: Kanaya K, Ishikawa A, Yaoita M, Niihori T, Aoki Y, Iba K, Yamashita T. Metachondromatosis without enchondromas: a case report and review of the literature. JBJS Case Connect, 2016 Apr 27;6(2):e30.
Metachondromatosis is a rare, hereditary disorder involving the formation of enchondromas and osteochondromas that was first described by Maroteaux. The complete form of the disease is characterized by multiple metaphyseal juxta-epiphyseal exostoses, metaphyseal enchondromas, periarticular calcifications, and frequently unilateral or bilateral Legg-Calvé-Perthes-like changes in the femoral head resembling osteonecrosis. Osteochondromas in metachondromatosis are true osteochondromas with cartilaginous caps, but characteristically point toward the adjacent joint, often involve the bones of the hands and feet, and do not produce shortening or growth disturbance of the affected bone. In contrast, enchondromas in metachondromatosis may appear as metaphyseal enchondromas. As far as we know, there has been no previously reported case of metachondromatosis without enchondromatous lesions.
In the present case, a full radiographic survey of the skeleton failed to demonstrate enchondromatous lesions. Furthermore, MRI failed to reveal any enchondromatous lesions in the pelvis or the proximal aspect of either femur, which are the areas most frequently affected by enchondromas. However, the osteochondromas in the present case showed characteristic features of metachondromatosis in that they pointed toward the adjacent joint, involved the bones of the hands, did not produce shortening or growth disturbance of the affected bone, and resolved spontaneously. The patient’s mother had also been affected on both hands in her youth, although her tumors resolved spontaneously.
Recently, heterozygous mutations in the PTPN11 gene were identified in patients with metachondromatosis. PTPN11 is a well-established human oncogene, and mutations in PTPN11 cause Noonan syndrome or juvenile myelomyelogenous leukemia as well as other leukemias and some solid tumors. Patients with metachondromatosis often carry a heterozygous loss-of-function mutation in the PTPN11 gene, which was identified in >50% of patients. As a heterozygous mutation in PTPN11, PTPN11c.1093-1G>A, was found in the present case, the patient was diagnosed with metachondromatosis despite the absence of enchondromatous lesions. Although PTPN11c.1093-1G>A is a novel causative mutation for metachondromatosis, PTPN11c.1093-1G>T is a known causative mutation. Therefore, it is possible that PTPN11c.1093-1G>A is a causative mutation for a variant of metachondromatosis in which there is a lack of enchondromas.
Vining et al. reported that three members of a single family, a father and two offspring, had multiple clinical and radiographic features, such as multiple osteochondromas pointed toward the adjacent joint and multiple enchondromas involving the pelvis and the proximal aspect of the femur, compatible with a diagnosis of metachondromatosis as well as with hereditary multiple osteochondromas. However, the father underwent sequencing of the EXT1 and EXT2 genes, revealing an EXT2 mutation. The authors suggested that the radiographic and clinical diagnosis of hereditary multiple osteochondromas and metachondromatosis might not be as reliable as previously assumed.
The present case appeared different from that of the family reported by Vining et al. in that multiple osteochondromas with a tendency for spontaneous resolution were mainly found on the hands, without any enchondromatous lesions on the body. However, in terms of the radiographic and clinical features, the case might demonstrate a kind of overlap between metachondromatosis and hereditary multiple osteochondromatosis similar to that described by Vining et al. In the present case, RT-PCR analysis was performed to detect genetic mutations in PTPN11, KRAS, RAF1, and SHOC2 that are causative of Noonan syndrome and metachondromatosis, Noonan syndrome, Noonan syndrome, and Noonan-like syndrome, respectively. Unfortunately, genetic analysis of EXT was not performed in our patient.
The most common type of conventional cartilaginous tumor is osteochondroma, also known as secondary peripheral chondrosarcoma, and the majority of such tumors harbor mutations in EXT1 or EXT2. The other major types are central and periosteal tumors, a substantial proportion of which harbor IDH1 or IDH2 mutations. In contrast, metachondromatosis, which develops as multiple lesions and has been linked to mutations in PTPN11, is a rare subtype of conventional cartilaginous tumor. As these disease entities may have overlapping phenotypes, recognition of the associated mutations will allow robust disease classification that can be exploited for patient diagnosis and treatment.
This report has some limitations. First, most but not all of the skeletal structures were examined. As osteochondromas are benign lesions, we hesitated to expose the patient to a full radiographic survey of his entire body. Second, there is a possibility that enchondromatous lesions had already resolved prior to the time of the survey. Herman et al. reported a case of metachondromatosis in which the enchondromas resolved spontaneously at the age of twenty-six years, although radiographs of the patient made at seven years demonstrated short, linear, striated enchondromatous lesions in the third metacarpal and several proximal phalanges.
In summary, we report on a case of metachondromatosis without any detectable enchondromatous lesions. Metachondromatosis may be suspected despite the absence of enchondromatous lesions when characteristic features of osteochondromas, an orientation pointing toward the adjacent joint or spontaneous resolution, are recognized. This fact suggests that the phenotypes of metachondromatosis are both multiple and variable.
Reference: Kanaya K, Ishikawa A, Yaoita M, Niihori T, Aoki Y, Iba K, Yamashita T. Metachondromatosis without enchondromas: a case report and review of the literature. JBJS Case Connect, 2016 Apr 27;6(2):e30.
What is the diagnosis?
Hereditary multiple osteochondromas
Ollier disease
Metachondromatosis
Chronic recurrent multifocal osteomyelitis
Multiple periosteal chondromas

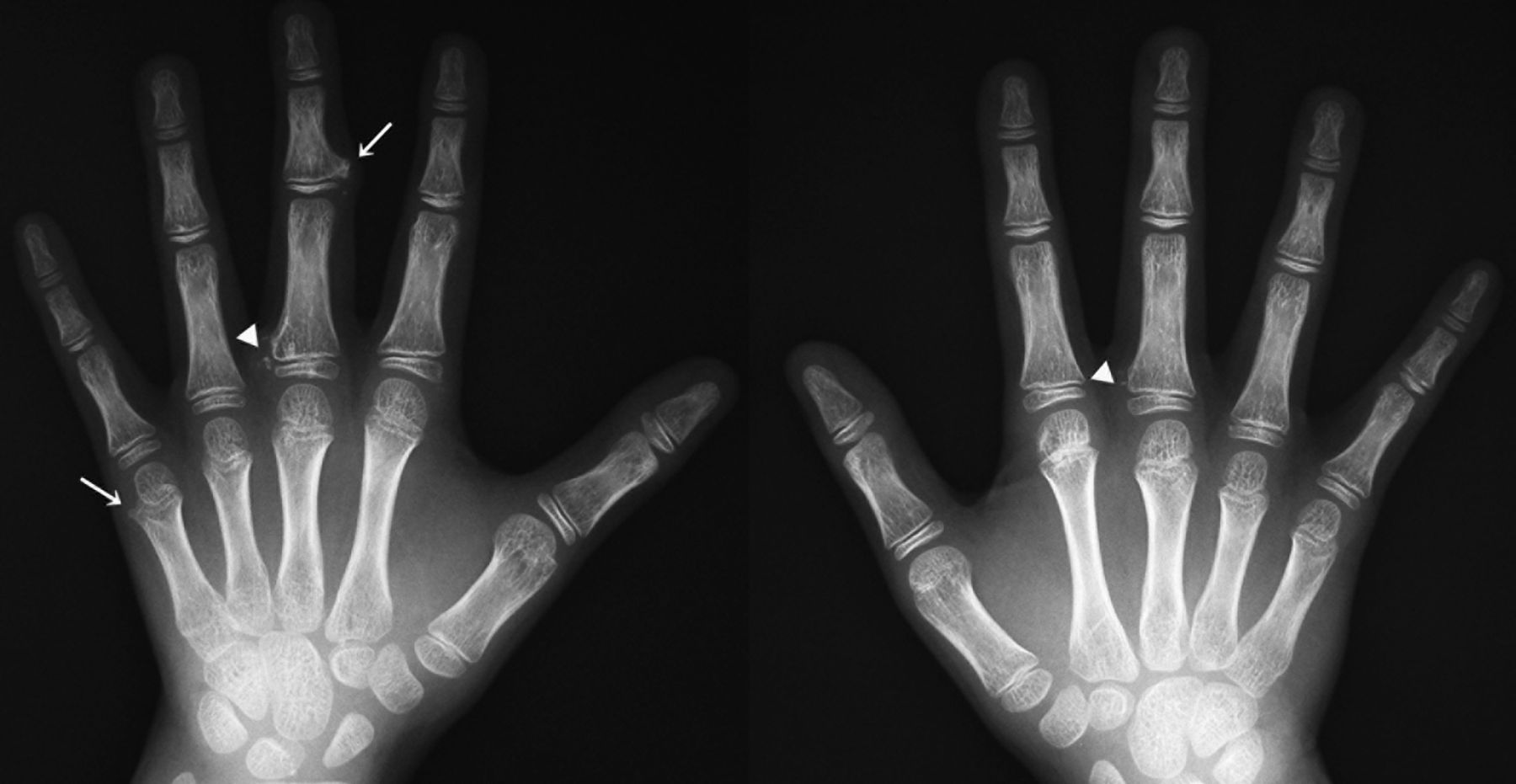
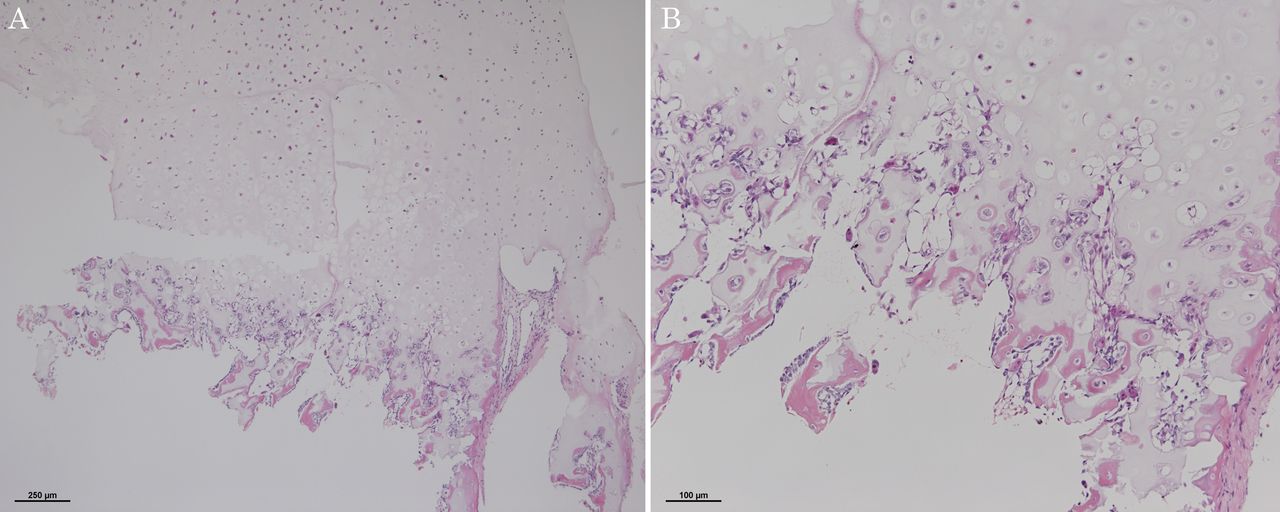
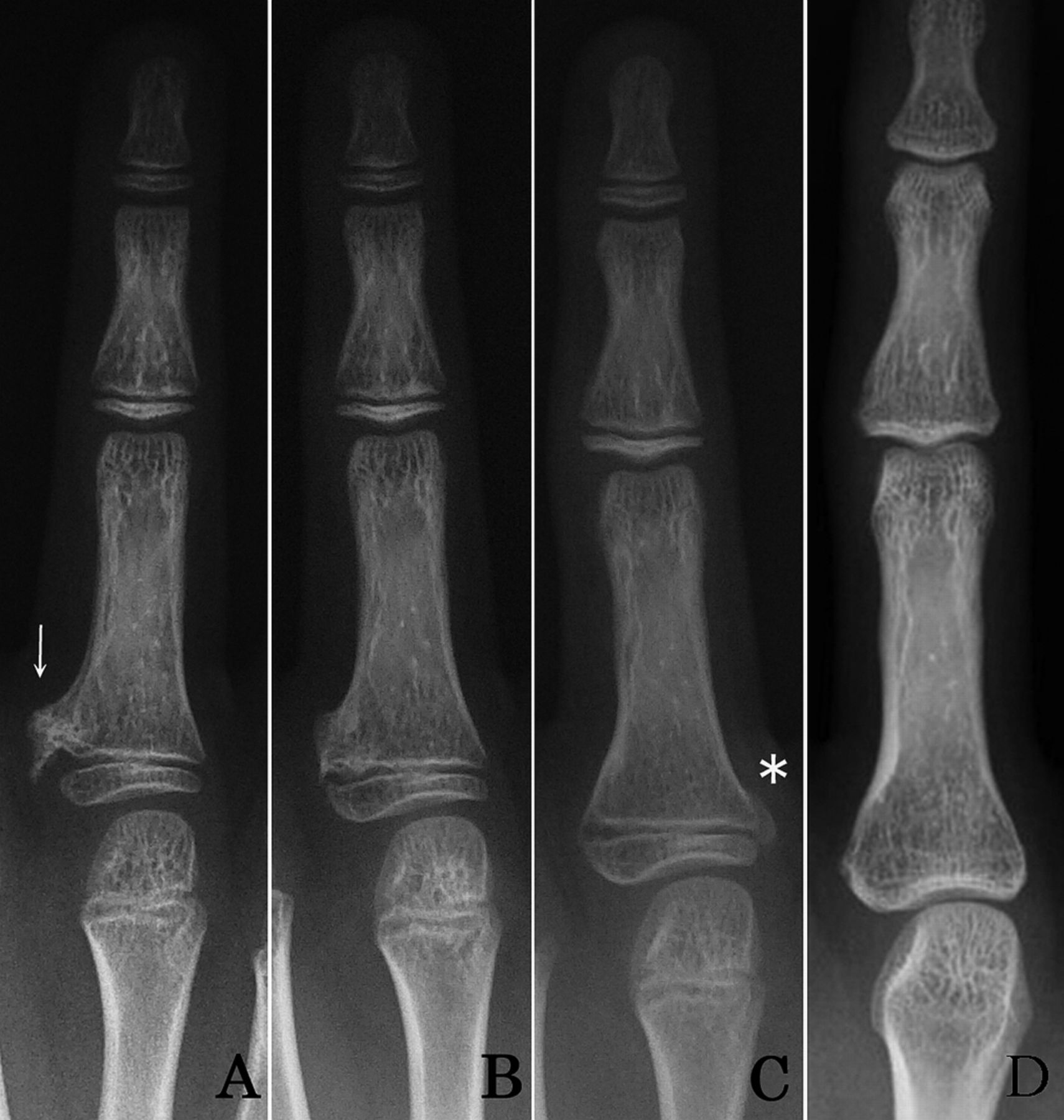
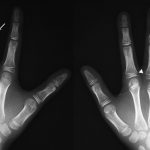 Fig. 1
Fig. 1![Fig. 2 Fig. 2 Histopathological sections of the osteochondroma from the proximal phalanx of the left middle finger showing a well-formed cartilage cap on the surface with enchondral ossification (hematoxylin and eosin, ×4 [Fig. 2-A] and ×10 [Fig. 2-B]).](https://quiz.jbjs.org/wp-content/uploads/2016/08/iqaug16-2_fig2-150x150.jpg) Fig. 2
Fig. 2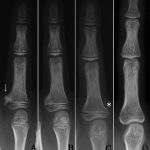 Fig. 3
Fig. 3 Fig. 4
Fig. 4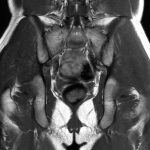 Fig. 5
Fig. 5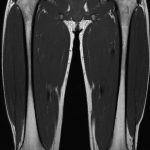 Fig. 6
Fig. 6