A Nine-Year-Old Girl with Ankle Pain and Swelling
August 3, 2016
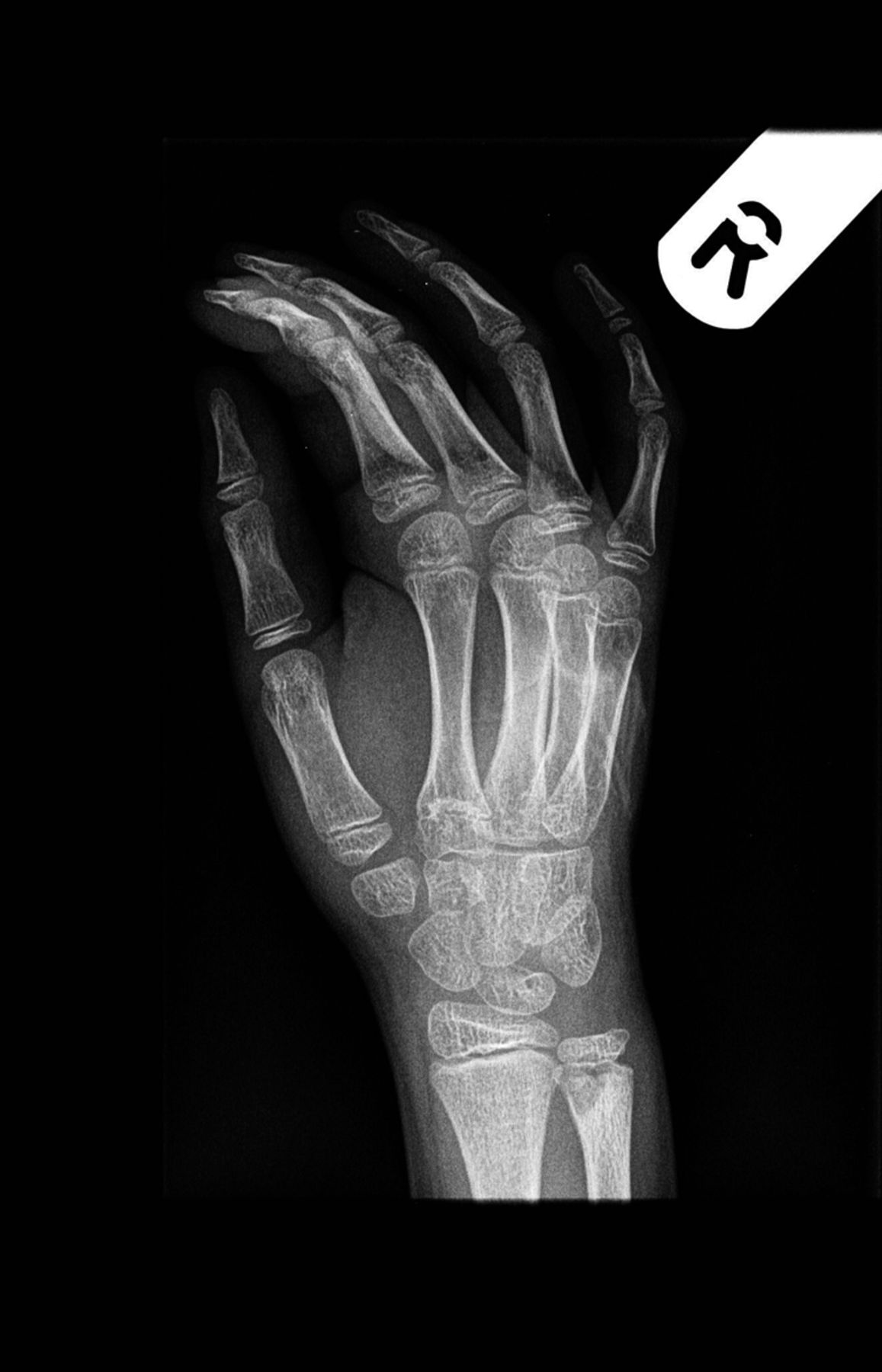
A 9-year-old Caucasian girl presented with a 3-week history of left ankle pain and swelling. Radiographs (Fig. 1) showed a lesion in the distal aspect of the fibula. The initial differential diagnoses included acute infection and tumor. Blood tests revealed a normal CRP (C-reactive protein) level. MRI (magnetic resonance imaging) was obtained (Fig. 2). She was treated with 2 weeks of intravenous ceftriaxone followed by 4 weeks of oral cefradine. Following antibiotic therapy, all blood parameters remained normal (CRP, 0.7 mg/L). Apart from a painless limp, there was symptomatic and radiographic improvement. Seven months later, she presented with similar symptoms in the contralateral ankle. Blood tests showed a marginally elevated CRP of 13 mg/L. Imaging (Fig. 3) showed similar lesions around the distal fibular and tibial epiphyses. She was treated with a longer period of both intravenous and oral antibiotics. Early symptomatic improvement was followed by pain and swelling despite antibiotic treatment.
In view of the nature of the disease and the failure to respond to antibiotics, NSAID (nonsteroidal anti-inflammatory drug) treatment in the form of ibuprofen (10 mg/kg thrice daily) was initiated, with a good response. A review 1 month after treatment showed symptomatic improvement in pain and swelling, indicating remission, and treatment was discontinued. Following 3 months of remission, she presented with pain and swelling of the proximal aspect of the left tibia and inability to bear weight. Radiographs (Fig. 4) showed lesions in the proximal tibial metaphysis, similar to those seen previously in the ankle. Further treatment with the same NSAID was successful and she resumed a normal pain-free gait.
A diagnosis of CRMO (chronic recurrent multifocal osteomyelitis) or SAPHO (synovitis, acne, pustulosis, hyperostosis, osteitis) was made although she had no dermatological manifestations. Two months later, she presented with a painful and swollen wrist. Radiographs demonstrated an osteolytic lesion in the distal aspect of the radius and ulna (Fig. 5), similar to the previous proximal tibial and ankle lesions. This was again treated successfully with the same NSAID.
Following this episode, she was treated with a 3-day course of pamidronate (0.5 mg/kg) on the advice of pediatric rheumatologists. The symptoms resolved for nearly 2 years. More recently, she was clinically evaluated for further episodes of pain in multiple joints; after appropriate investigations, she was treated with NSAIDs (naproxen, 125 to 250 mg twice daily), with complete resolution of symptoms.
Proceed to Discussion >>Reference: Mallick A, Chaturvedi A, Muthukumar N. Chronic recurrent multifocal osteomyelitis: a diagnostic dilemma: a case report and review of the literature. JBJS Case Connect. 2016 Jun 08;6(2):e43.
The acronym CRMO is misleading as this disorder is not necessarily chronic, recurrent, or multifocal. It is inflammatory rather than infective, and is part of a clinical spectrum of disorders collectively known as chronic nonbacterial osteitis (CNO or NBO). Other disorders in this group are SAPHO syndrome and diffuse sclerosing osteomyelitis (DSO) affecting the mandible. Osteomyelitis itself is a vague term, essentially inflammation of bone, usually due to infection but often with no identifiable pathogen.
The pathogenesis of CRMO is poorly understood. CRMO was originally attributed to an infectious pathology because of its clinical, histological, and radiographic similarities to bacterial osteomyelitis. If bacteria are identified from an inflammatory bone lesion, then treatment with antibiotics is appropriate. However, failure to isolate a specific organism and lack of response to antibiotic therapy make a bacterial cause increasingly unlikely.
There have been reports of CRMO associated with bacterial isolates, in particular Propionibacterium acnes affecting the spine and sternum. This organism is an aerotolerant gram-positive anaerobe that is commonly associated with facial acne and is sensitive to a wide range of antibiotics. It is worth keeping in mind that P. acnes is a commensal organism in the skin, and that any bone biopsy may also include some skin along with the bone. This low-virulence organism may potentially be implicated in the etiology of CRMO in some instances, by acting as an antigenic trigger of bone marrow inflammation. P. acnes is thought to activate toll-like receptors (TLRs), including TLR9, TLR2, and TLR4, which lead to an imbalance of pro-inflammatory and anti-inflammatory cytokines. P. acnes is also commonly interpreted as a contaminant when detected in microbiological cultures, so its role in CRMO is unclear.
In patients with active CRMO, the level of TNF (tumor necrosis factor)-alpha has been reported to rise locally and systemically, suggesting a possible immune-mediated etiology. There appears to be a genetic link, as cases have been reported in siblings, including monozygotic twins. Supporting a genetic link is the identification of the LPIN2 gene in Majeed syndrome, an autosomal-recessive disorder with bone lesions similar to those in CRMO. The LPIN2 gene is thought to play a role in regulating innate immunity.
Other genes associated with CRMO include PSTIP1 and PSTIP2, mutations in which are thought to result in “pyogenic arthritis, pyoderma gangrenosum, and acne syndrome” (PAPA syndrome) and SAPHO syndrome, respectively. Mutations in the NOD2 gene are associated with other autoinflammatory conditions such as inflammatory bowel diseases and Blau syndrome. Mutations in the IL1RN gene have been discovered in patients with neonatal onset of sterile multifocal osteomyelitis, periostitis, and pustulosis. Most of these autoinflammatory conditions include dermal and osseous as well as other systemic involvement.
CRMO is recognized worldwide and has been reported to account for 2% to 5% of cases initially presumed to represent osteomyelitis. It has a 2:1 female predominance. Mean age of onset is 8.5 years. Most cases go into remission in late childhood; some may relapse in late teenage years or early adulthood.
CRMO is difficult to diagnose, in part because of its variable presentation and broad differential diagnoses, which include infection, primary benign or malignant osseous neoplasms, lymphoma, metastasis, eosinophilic granuloma, and fibrous dysplasia.
The osseous lesions in CRMO usually occur sequentially, predominantly involve the metaphysis of long bones, and are frequently relapsing. They can also involve the clavicle, sternum, vertebral bodies, and pelvis, mainly the sacroiliac joints. There are asymptomatic periods punctuated with flare-ups. The painful osteoarticular focal lesions of CRMO cause swelling and erythema of the overlying tissue, as would be expected with an inflammatory process.
Between 30% and 80% of patients with CRMO will develop arthritis, including spondyloarthropathy, in their lifetime. Associations with inflammatory bowel disease and psoriatic arthritis have been reported, as patients have been found to have mutations in genes related to those conditions (IBD1 and PSORAS1, which are now known to be the same as the NOD2 gene).
Aside from the osteoarticular predilection, there can be involvement of other systems and organs, including the skin, eyes, gastrointestinal system, and lungs. Dermatological manifestations, the most common extra-osteoarticular features, include palmoplantar pustulosis, psoriasis, and occasionally pyoderma gangrenosum. This is of special interest as most of the skin manifestations of CNO/CRMO also occur in multiple other autoinflammatory disorders.
King et al. proposed that, to make a diagnosis of CRMO, all of the following criteria should be met: (1) multifocal bone lesions, (2) prolonged clinical course with remissions and relapses, (3) lack of response to antimicrobial therapy, (4) radiographic examination demonstrating multifocal bone lesions showing osteolysis surrounded by sclerosis, and (5) lack of identifiable etiology.
Our patient met all of the above criteria, as she presented with multifocal lesions, a prolonged clinical course with remission and recurrences, and a lack of a predictable and consistent response to antibiotics.
Jansson et al. proposed a diagnostic aid for NBO with major and minor criteria. According to that classification, NBO (of which CRMO is considered a subtype) is confirmed by the presence of 2 major criteria or of 1 major and 3 minor criteria.
Laboratory parameters are nonspecific in CRMO. The leucocyte count ranges from 8 to 9.6 × 10/L, the ESR (erythrocyte sedimentation rate) ranges from 11 to 20 mm/hr, and the CRP level ranges from 1.4 to 15.6 mg/L, depending on recurrence and the number of foci. Radiographic appearances are variable and can be sclerotic, lytic, or mixed. MRI can be helpful in diagnosis by differentiating neoplastic and infectious conditions from inflammatory features of CRMO, although there are no findings that can be considered pathognomonic for the latter condition. Gikas et al. and Depasquale et al. suggest that an experienced musculoskeletal radiologist can help distinguish CRMO from other pathologies on the basis of imaging by utilizing MRI and that a biopsy should not be performed routinely. Features of lytic destruction adjacent to the growth plate and demarcated by a sclerotic rim, without periosteal new bone formation or sequestra, are characteristic of CRMO early in its course. Bone marrow involvement in particular is better demonstrated on MRI than on radiographs. There may be decreased signal on T1-weighted images and scattered areas of increased signal on T2-weighted images. Absence of soft-tissue inflammation, of abscess formation, and of sinus tract formation on MRI distinguishes CRMO from sarcomas and infective osteomyelitis. Whole-body MRI is being used in some centers at the start of management to characterize CRMO lesions at their onset, evaluate treatment response, and correlate radiographic findings with clinical response. An isotopic bone scan is also useful for identifying sites affected.
Diagnosis can be difficult when only 1 site is involved. In such cases, a biopsy can be performed to exclude neoplastic and infectious pathologies. Biopsy and histological examination can support the diagnosis but are not specific. Histologically, CRMO shares many features with acute osteomyelitis: scattered granulocytic inflammatory infiltrates within affected lesions. The infiltrates consist predominantly of lymphocytes, plasma cells, histiocytes, and granulocytes. Synovial biopsy samples from adjacent joints may also demonstrate lymphocytic and granulocytic infiltrates. Aseptic osteitis is the hallmark. A negative culture, however, cannot definitively rule out an infective etiology.
Management of CRMO is focused on anti-inflammatory and immunosuppressive treatment. Traditionally, the aim of management is symptom control and the prevention of relapses. Antibiotics are frequently used prior to a diagnosis to cover possible bacterial osteomyelitis. Assmann et al. followed patients with SAPHO treated with antibiotics. These seemed to offer temporary control, but relapse was observed on cessation. Other studies have also suggested that the temporary benefit can be attributed to the anti-inflammatory and immunomodulatory effects rather than the antimicrobial properties of antibiotics.
Some authors recommend NSAIDs as a first-line treatment for acute attacks and relapses. For example, treatment with NSAIDs showed favorable response in a study by Beck et al., with 67% of cases becoming completely clinically silent after treatment.
Patients with frequent relapses despite NSAIDs are commenced on disease-modifying anti-rheumatic drugs (DMARDs). Corticosteroid therapy can be used for short periods as an adjunct, along with NSAIDs, while DMARDs are being started.
Bisphosphonates, with their cytokine-suppressive properties, have also shown promise and are reserved for cases refractory to NSAIDs and DMARDs. Pamidronate is the most commonly used bisphosphonate. It is particularly useful in patients with vertebral involvement. Up to 40% of patients with vertebral CNO have associated crush fractures, and vertebral remodeling after pamidronate treatment has been reported. Current reports suggest that approximately 70% of patients with CNO have long-lasting symptom control following pamidronate treatment.
The prognosis in cases of CNO is generally favorable, with the resolution of symptoms observed in most patients as they reach adulthood. Longer-term studies have shown that up to one-quarter of patients do have residual disease, which can lead to osseous deformity and a poorer quality of life or painless limp. However, >75% had minimal or no physical disability.
Catalano-Pons et al. reviewed the clinical, biological, and radiographic data of children with CRMO at 5 French pediatric centers and found that over one-half (58.6%) of patients had active disease at some time during follow-up (at 6 months to 15 years following diagnosis), although a minority of patients had a severe and prolonged disease course despite intensive treatments.
Although CRMO is considered rare, the prevalence is likely to be underestimated because of poor recognition of the disease. The diagnosis of CRMO is hampered by lack of a specific diagnostic test. Although algorithms for clinical diagnosis have been proposed in the literature, no one study has the case numbers necessary to validate an algorithm for extended clinical use.
We propose that, as soon as a suspicion of CRMO is raised, MRI scanning should be undertaken and reviewed by musculoskeletal radiologists, perhaps in pediatric, bone tumor, or bone infection centers. A biopsy should not be necessary in most cases. The balance of probability and clinical suspicion—based on clinical assessment and available radiographic and serological examinations—should be relied on to limit surgical morbidity where possible. Follow-up studies of larger patient cohorts are warranted to improve diagnosis and management of CRMO.
Reference: Mallick A, Chaturvedi A, Muthukumar N. Chronic recurrent multifocal osteomyelitis: a diagnostic dilemma: a case report and review of the literature. JBJS Case Connect. 2016 Jun 08;6(2):e43.
What is the diagnosis?
Bacterial osteomyelitis
Polyostotic fibrous dysplasia
Langerhans cell histiocytosis
Chronic recurrent multifocal osteomyelitis
Peripheral T-cell lymphoma

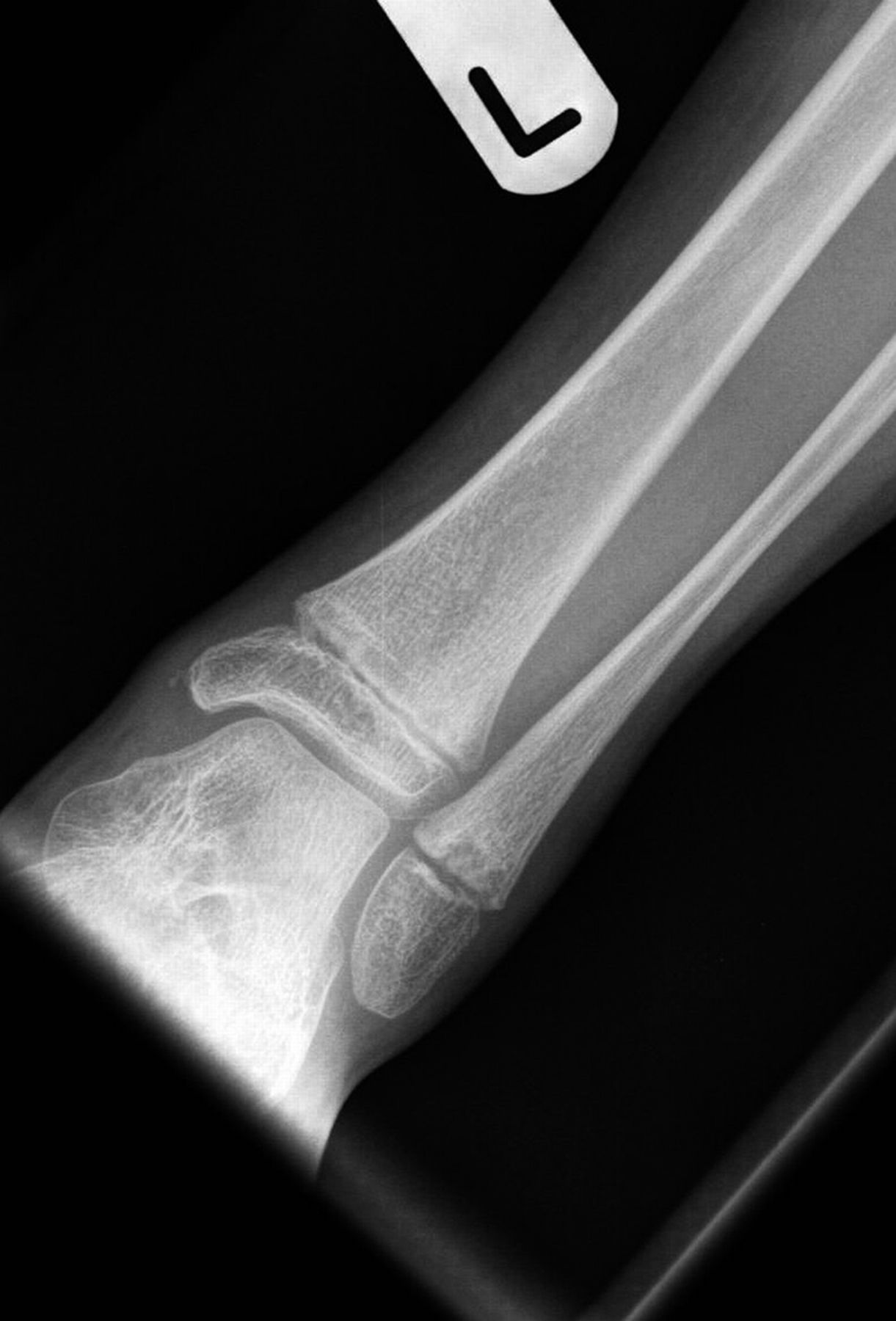

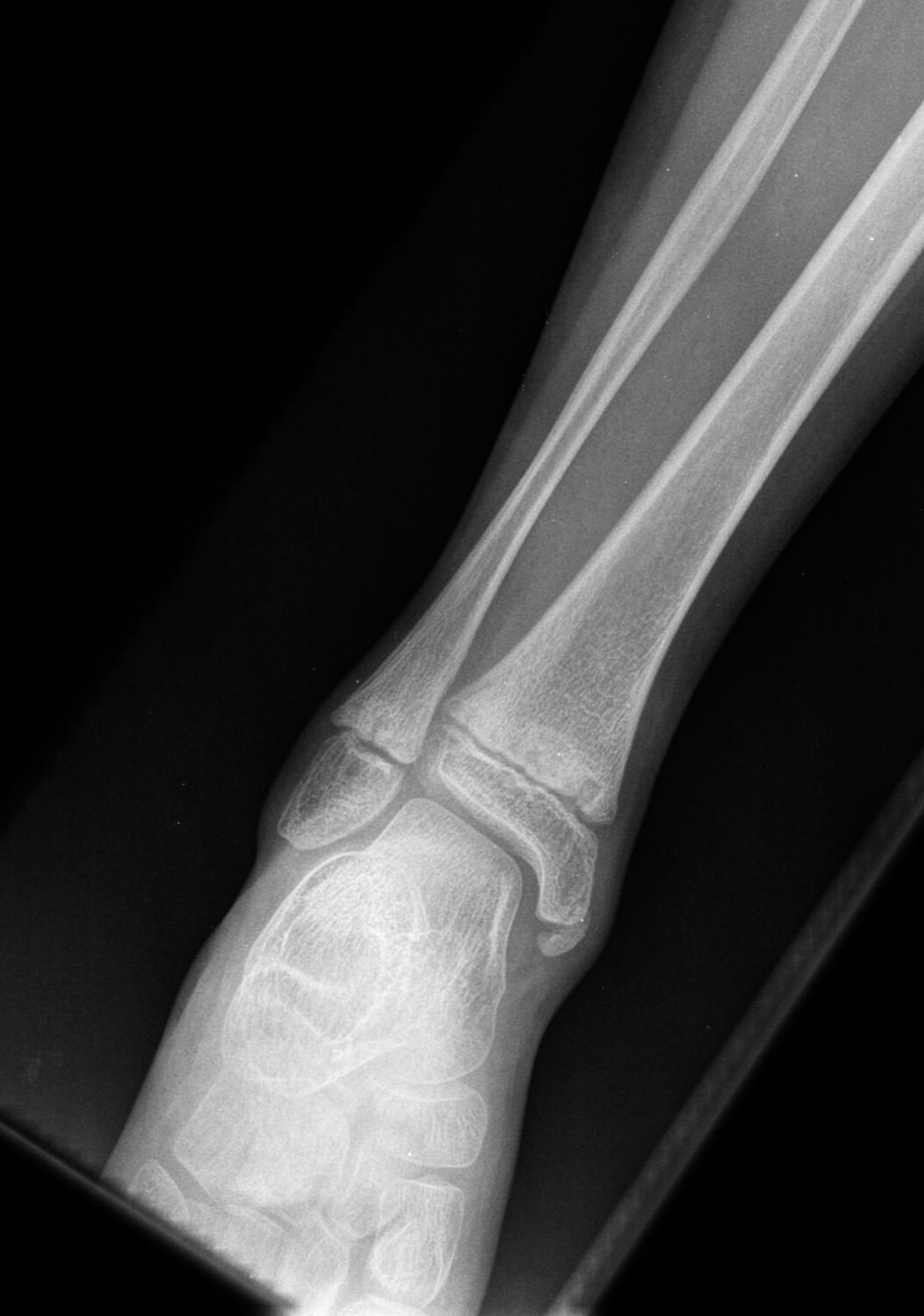
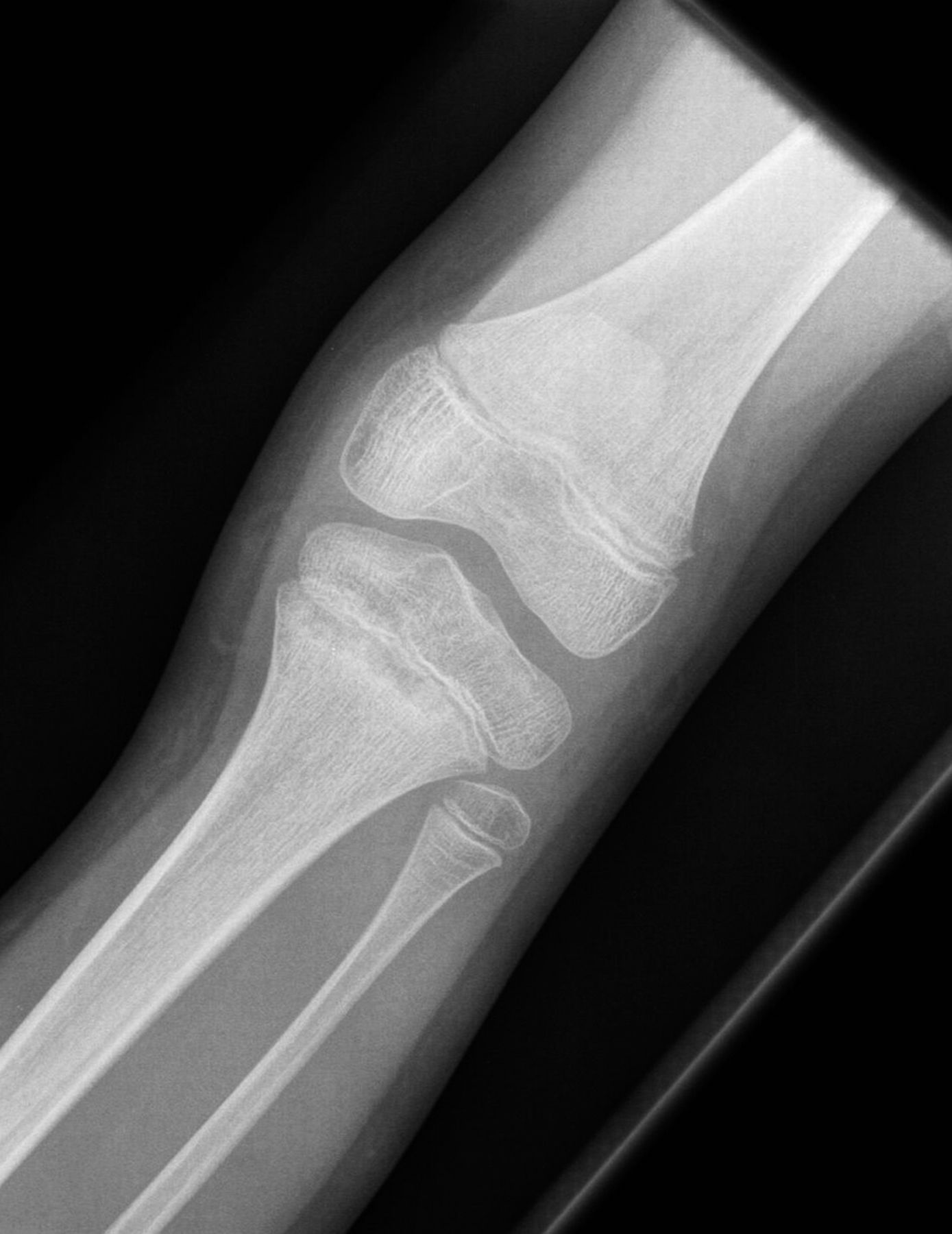
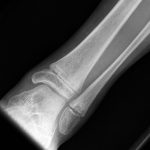 Fig. 1
Fig. 1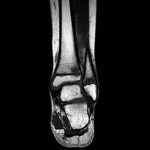 Fig. 2
Fig. 2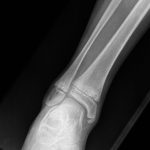 Fig. 3
Fig. 3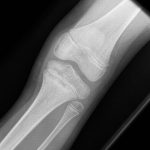 Fig. 4
Fig. 4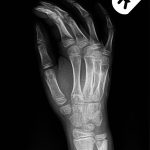 Fig. 5
Fig. 5